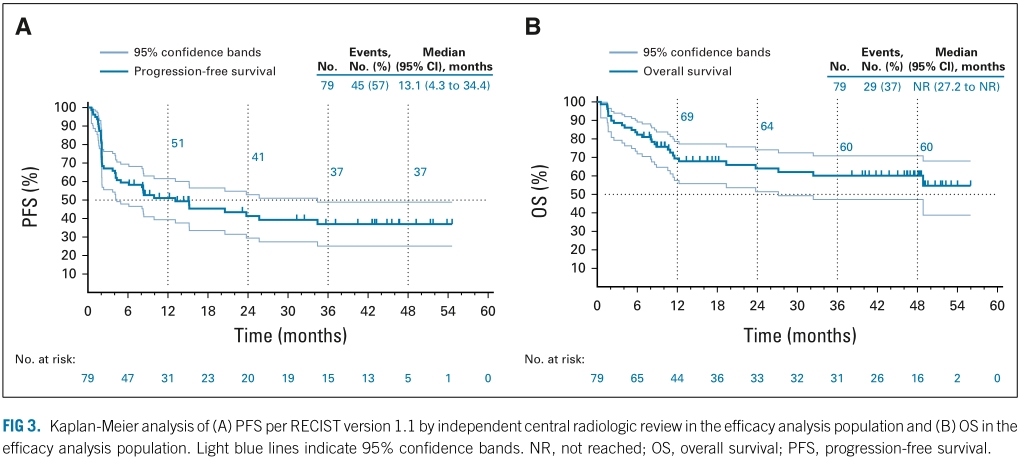
Bariani: Financial Interests, Personal, Funding: Mabxience Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA Bristol Myers Squibb Financial Interests, Personal, Advisory Role: Libbs. O'Malley: Financial Interests, Personal, Advisory Board: AstraZeneca, Tesaro/GSK, BBI, Immunogen, Ambry, Janssen/J&J, Abbvie, Regeneron, Amgen, Novocure, Genentech/Roche, GOGFoundation, Iovance Biotherapeutics, Inc, Myriad Genetics, Eisai, Agenus, Tarveda, Merck & Co., Inc., Kenilworth, NJ, USA, SeaGen, Novart Financial Interests, Institutional, Funding: AstraZeneca, Tesaro/GSK, Immunogen, Janssen/J&J, Abbvie, Regeneron, Amgen, Novocure, Genentech/Roche, VentiRx, Array Biopharma, EMD Serono, Ergomed, Ajinomoto Inc., Ludwig Cancer Research, Stemcentrx, Inc, Cerulean Pharma, GOGFoundation, NCI, Bristol Mye. Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. Writing support was provided by Christabel Wilson, MSc, of ICON plc (North Wales, PA, USA), funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. “+” indicates there was no PD by the time of last disease assessment. Pembro demonstrated robust and durable ORR (48% CR, 14%), encouraging survival outcomes and manageable toxicity in pts with heavily pretreated, advanced MSI-H/dMMR EC, and is a promising treatment option in this setting. 25 pts (28%) had immune-mediated AEs and infusion reactions (grade 3–4, 7% no grade 5). 68/90 pts (76%) had treatment-related AEs (grade 3–4, 12% no grade 5), and led to discontinuation in 7%. ORR was 48%, with DOR ≥3 y in 68% see the table for results. In the efficacy population (n = 79), median time from first dose to data cutoff was 42.6 (range, 6.4–56.1) mo 41 (52%) had ≥2 lines of prior therapy and 61% had ECOG PS 1. ResultsĪs of Oct 5, 2020, 18/90 pts (20%) had completed 35 cycles of pembro and 52 (58%) had discontinued treatment. Efficacy was assessed in pts who received ≥1 pembro dose and had ≥26 wk follow up and safety in all treated pts. Secondary endpoints included DOR, PFS, OS, and safety. 
Primary endpoint was ORR per RECIST v1.1 by independent central radiologic review. Pts received pembro 200 mg Q3W for up to 35 cycles. In cohort K, MSI-H/dMMR output was determined prospectively by PCR and/or IHC at a local lab, respectively. In cohort D, MSI-H expression was determined retrospectively by PCR at a central lab. Pts from cohorts D (EC, regardless of MSI-H status) and K (any MSI-H/dMMR solid tumor, except colorectal) were ≥18 y, had previously treated, advanced MSI-H/dMMR EC, measurable disease per RECIST v1.1, ECOG PS 0/1 and provided a tumor sample for biomarker analysis. 
We present longer follow up in more pts from KEYNOTE-158. ORR of 57% was observed in 49 pts with MSI-H/dMMR EC in KEYNOTE-158. Pembro showed durable and clinically meaningful benefit in previously treated, advanced MSI-H/dMMR tumors, including EC, in the nonrandomized, open-label, phase II KEYNOTE-158 study (NCT02628067). Treatment options for previously treated, advanced MSI-H or mismatch repair deficient (dMMR) tumors are limited. 13 Division Of Medical Oncology And Immunotherapy, Center for Immuno-Oncology, Department of Oncology, University Hospital of Siena, Siena/IT.12 Oncology, Merck & Co., Inc., Kenilworth/US.11 Biostatistics, Merck & Co., Inc., Kenilworth/US.10 Department Of Medical Oncology, Peter MacCallum Cancer Centre, Melbourne/AU.9 Early Phase Trials And Sarcoma Units, Institut Bergonie, Bordeaux/FR.8 Oncology Department, Tel Aviv Medical Center, Tel Aviv and Sackler School of Medicine, Tel Aviv University, Tel Aviv/IL.7 Segal Cancer Centre, Jewish General Hospital, Rossy Cancer Network and Departments of Oncology and Medicine, McGill University, Montreal/CA.6 Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins, Baltimore/US.5 Division Of Medical Oncology, Princess Margaret Cancer Centre, Toronto/CA.4 Drug Development Department, Gustave Roussy, Institut National de la Santé et de la Recherche Médicale U1015, Villejuif/FR.3 Department Of Medical Oncology, Centre Léon Bérard, Lyon/FR.2 Department Of Medical Oncology, Instituto do Câncer do Estado de São Paulo, Universidade de São Paulo, São Paulo/BR.1 Division Of Gynecologic Oncology, The Ohio State University Wexner Medical Center and The James Comprehensive Cancer Center, 43210 - Columbus/US.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
